Built for a business world in constant motion and driven by scientific innovation, LabVantage LIMS platform expands to suit your lab’s changing needs and your company’s evolving position. With advanced analytics to generate value from your scientific data lake, an all-in-one platform to simplify your tech stack and unburden your IT department, and an intuitive interface to accelerate adoption, you’ll love working with the most modern LIMS available—today, and far into the future.
Configure your no-code LIMS quickly and easily, or take advantage of pre-packaged LIMS accelerators tailored for your industry.
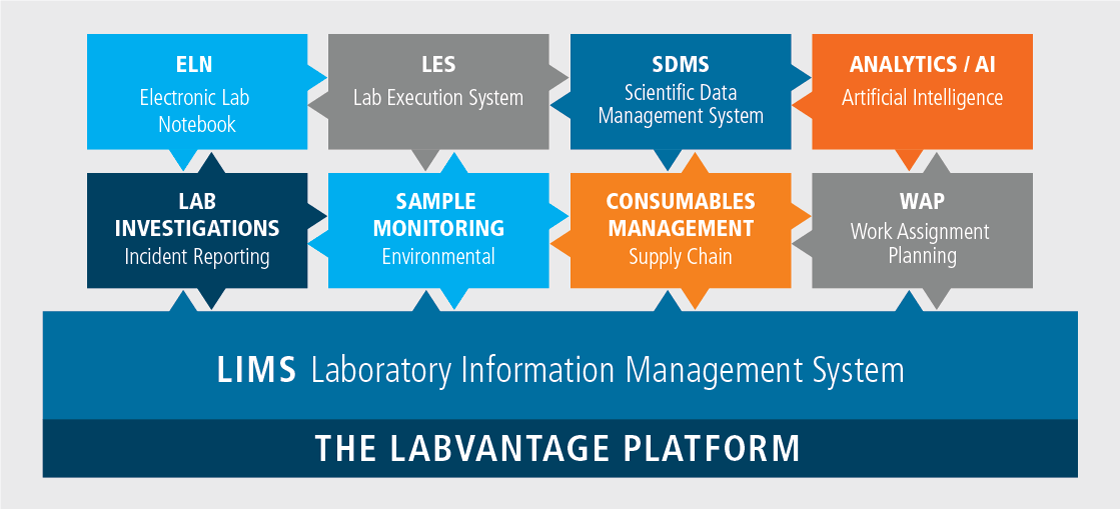
Grow your SaaS platform with flexible, on-demand access to integrated ELN, LES, and more through a single LIMS interface.
Establish your lab of the future by harnessing AI, leveraging your scientific data lake, and availing our scientific semantic search capabilities.
See how our Scientific Data Management System ensures compliance and data integrity.
Go under the hood and see the power of our browser-based, HTML5-compliant platform.
The overwhelmingly popular option is Software as a Service. With Enterprise and Standard SaaS offerings, LabVantage presents choices that eliminate upfront capital investment while maximizing your flexibility, uptime, and security.
Available in partnership with Amazon Web Services, cloud hosting reduces reliance on your internal IT resources.
Not yet ready for SaaS? We offer convenient on-premises deployment with consulting services to support your ongoing platform management.
