Pharmaceutical LIMS
The only pre-validated, pre-configured LIMS for Pharma and Biotech
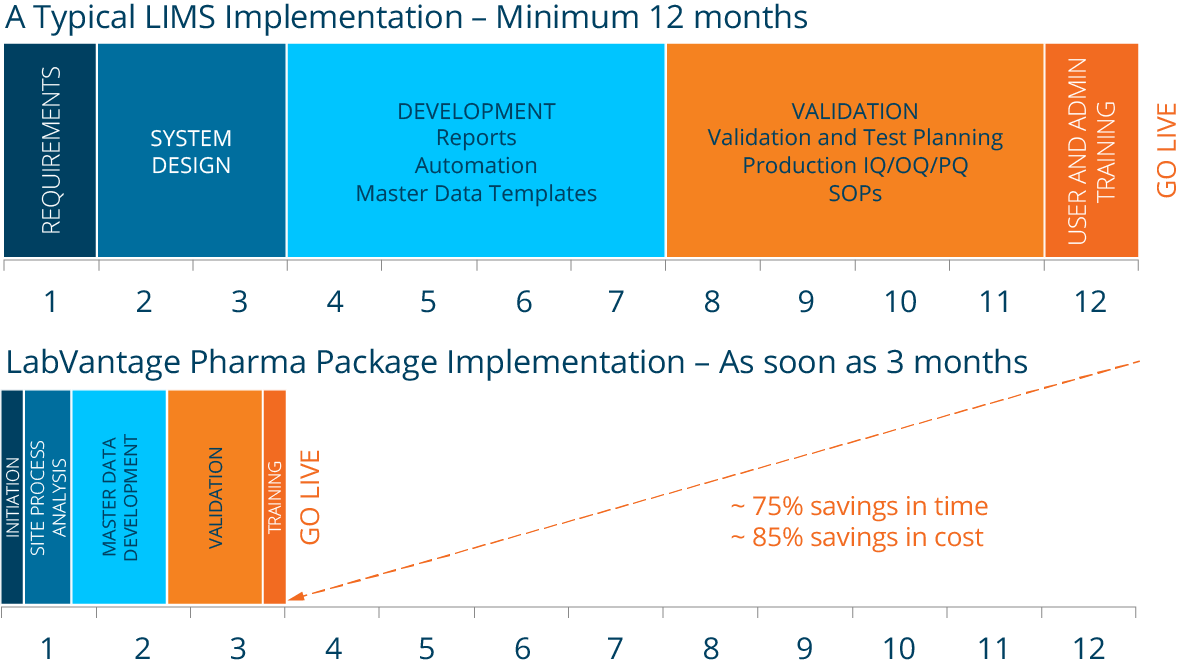
Deploy quickly, work productively with LabVantage Pharma LIMS
Imagine the immense time and cost savings from being able to deploy a full end-to-end informatics solution for your R&D, analytical, QC manufacturing, and other labs where the critical validation of the software as delivered was already done for you.
LabVantage provides the world’s only pre-validated and pre-configured pharmaceutical LIMS, allowing you to save up to 85% in implementation costs and 75% in time compared to a traditional LIMS implementation. The out-of-the box functionality includes everything you need, from batch management, stability testing, and consumables management to environmental monitoring, barcode label printing, and more.
Highly flexible and robust, LabVantage Pharma LIMS scales and supports your organization, whether you are monitoring quality control for launching your first product or collecting stability data on multiple products. Advanced functionality, too, like predictive and prescriptive analytics, helps digitally transform your organization, so you can streamline processes, boost productivity, and leverage valuable scientific and business insights from your data.
Count on LabVantage Pharma for:
- Out-of-the-box fully operational and functional, pre-validated LIMS pre-configured with pharma and biotech templates and workflows
- Pre-validation in compliance with GAMP 5 guidelines
- Compliance with FDA 21 CFR Part 11, European Union’s EudraLex Annex 11, and other global regulations
- Rapid, reduced-risk deployment, ideal for labs with limited IT resources
- Embedded ELN, LES, and SDMS for managing day-to-day operations, research, and data management – no point solutions to knit together
- Deploy via validated SaaS or on-premises
Manage your pharma R&D, Analytical, Quality, and other labs with precision and efficiency – with a single laboratory informatics platform.
Why choose LabVantage Pharma
One comprehensive integrated solution
Rather than bolted-together point solutions that can cause data discrepancies, take advantage of one comprehensive solution for greater data accuracy, improved end-to-end visibility, and easier collaboration among teams – from R&D to manufacturing.
Best practices
Purpose-built functionality for the pharma/biotech industry contains standardized workflows built-in so you can be assured your labs follow consistent processes, align with strict regulations, and follow top quality control standards for reliable results.
Future-Proof Your Business
Industry regulations and conditions change over time, requiring your lab to adapt if you want to stay relevant and grow. You might also add new service areas, new tests, or new users as you expand capabilities. LabVantage Pharma is highly flexible and easy to personalize for changing situations—with no specialized coding expertise needed. LabVantage is also built using the most technologically advanced and secure platform which is continually tested and enhanced to remain ahead of the technology curve.
Highly secure and accessible
LabVantage Pharma is 100% browser-based, HTML5-compliant, with zero-footprint user interface. This means the solution is highly secure, easy to access globally from one centrally hosted system, and can be personalized using LabVantage tools. Deployed via validated SaaS or on-premise – each covers multi-site, multi-location installations.
Expertise at your service
LabVantage understands your unique needs as no other solution provider does. We are experts in pharma and biotech lab information management with solution engineers, business analysts, and project managers worldwide to support you, from deployment through validation and ongoing monitoring. A majority of top 20 global pharma, biotech leaders, and emerging companies rely on LabVantage for lab informatics.
Data Auditing and Integrity Functionality
LabVantage Pharma includes dynamic auditing, a feature purpose-built to help pharma companies adhere to existing and proposed data integrity guidelines from global regulatory agencies. Labs will automatically maintain a complete and thorough GxP-compliant audit trail fully viewable in the LIMS.



